The Clinical Studies Behind Valeda Light Therapy: What the Research Says
If you’ve been diagnosed with dry AMD, you may hear about both AREDS2 vitamins and Valeda light therapy. They can sound similar because both are meant to help protect your vision, but they work in very different ways.
What the LIGHTSITE Trials Show
When a new treatment enters the world of retina care, the question every patient and physician should ask is the same: what does the evidence say?
For Valeda, the first FDA-authorized treatment proven to improve vision in dry AMD, the answer is grounded in a robust, multi-phase clinical program called the LIGHTSITE trials.
Here is a close look at what those studies found and why the results matter for patients with dry age-related macular degeneration.
The LIGHTSITE Program
LumiThera, the company behind Valeda, built the case for FDA authorization through a series of progressively larger and more rigorous clinical trials, each one building on the last. The program spans over a decade of research, beginning with early feasibility work and culminating in a landmark pivotal trial and long-term extension study.
LIGHTSITE I: Early Promise
LIGHTSITE I was the first clinical study to evaluate multiwavelength photobiomodulation (PBM) for dry AMD in a controlled setting.
The study enrolled 30 subjects (46 eyes) and followed them over 12 months. Even at this early stage, the results were encouraging:
- Patients treated with Valeda showed measurable improvements in best-corrected visual acuity (BCVA), contrast sensitivity, microperimetry, and quality of life.
- Researchers also observed reductions in central drusen volume and thickness, suggesting that PBM may be addressing underlying pathological changes associated with dry AMD.
These anatomical findings fueled significant scientific interest and laid the groundwork for larger trials.
LIGHTSITE II: Confirming the Signal
LIGHTSITE II was a double-masked, sham-controlled, parallel-design, prospective multi-site study that expanded the evaluation of PBM for dry AMD. The sham-controlled design was important, helping ensure that any observed benefits were attributable to the treatment itself and not a placebo effect. Results continued to support the safety and potential efficacy of Valeda, providing the confidence needed to move into the pivotal Phase III trial.
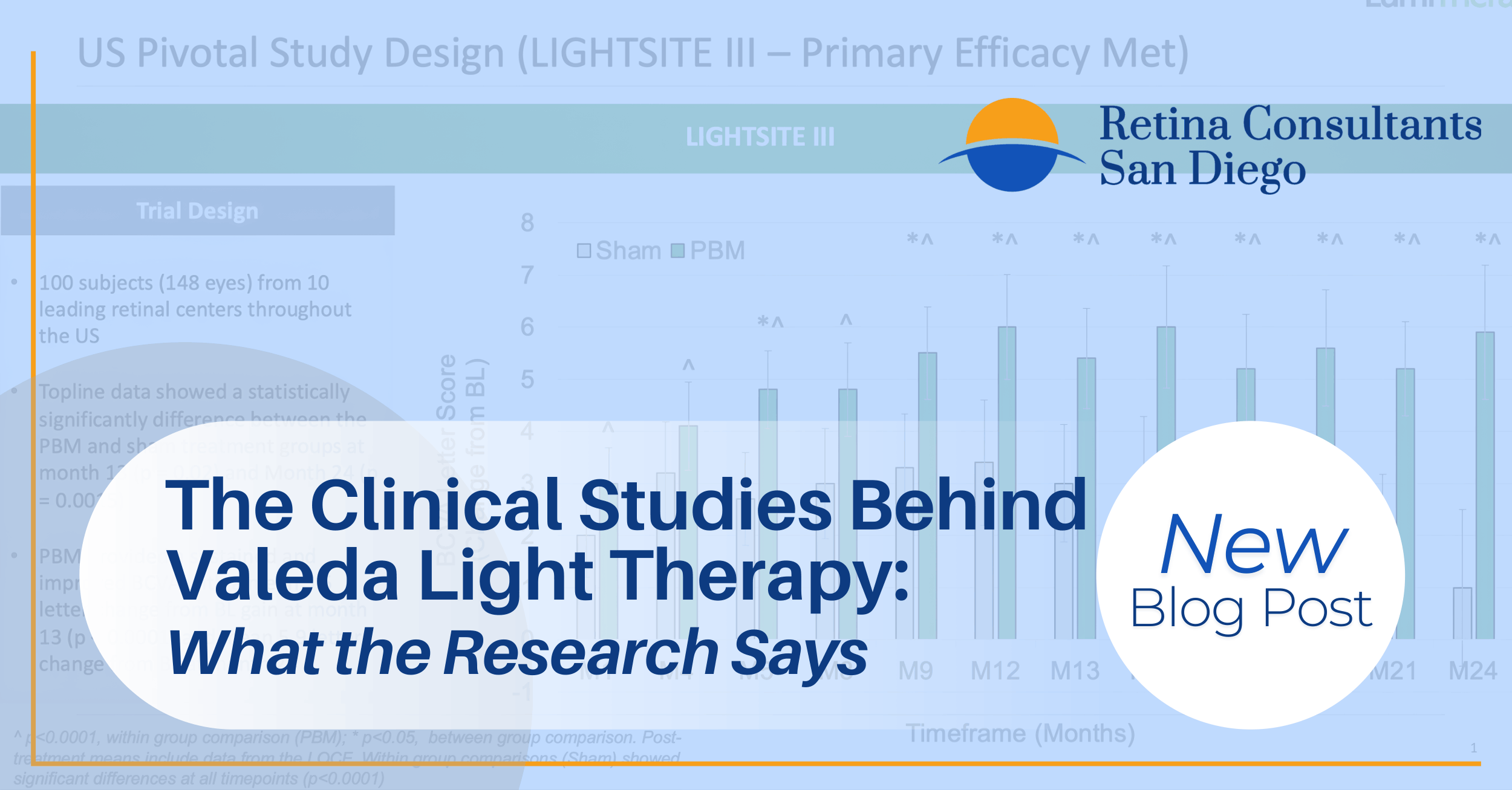
LIGHTSITE III: The Pivotal Trial
LIGHTSITE III is the cornerstone of the Valeda evidence base and the study that ultimately supported FDA authorization in November 2024. It was a double-masked, randomized, sham-controlled, parallel-group, multi-center trial conducted across ten U.S. clinical sites. The trial enrolled 100 subjects (148 eyes) with early to intermediate dry AMD, randomized in a 2:1 ratio to either active PBM treatment or sham treatment. Subjects received nine treatment sessions over three to five weeks, repeated every four months for a total of 24 months.
The key findings were significant:
- Valeda met the primary efficacy endpoint, demonstrating a statistically significant gain of 6.2 letters of best-corrected visual acuity at Month 21 (p = 0.0036), maintained at 5.6 letters at Month 24.
- The PBM group showed a 9.4 times lower odds of developing new-onset geographic atrophy compared to the sham group (p = 0.024).
- The treatment demonstrated an excellent safety profile with no serious device-related adverse events.
The combination of functional vision improvement and a measurable reduction in geographic atrophy progression suggests that Valeda may be doing more than just helping patients see better in the short term. It may be modifying the disease itself.
LIGHTSITE IIIB: Long-Term Extension
Following the conclusion of LIGHTSITE III, LumiThera conducted the LIGHTSITE IIIB Extension Trial, a prospective, open-label study that re-enrolled subjects from the original pivotal trial for an additional 13 months of Valeda treatment. A notable feature of this study was the gap between the two trials: subjects went approximately 20 months without treatment after completing LIGHTSITE III before resuming Valeda in the extension. Despite this prolonged pause, patients had retained meaningful vision gains from the original trial, and upon retreatment, many recovered back toward their prior peak.
Topline results presented at the ARVO 2025 annual meeting showed:
- More than 60% of subjects who received Valeda across both studies demonstrated a vision benefit of greater than one line on the eye chart.
- Follow-up data now extend to 4.5 years of observation and treatment exposure.
As Quan Dong Nguyen, MD, of Stanford University School of Medicine noted: “This is the first and only FDA-authorized treatment that can improve vision with extended benefits out to 4.5 years, suggesting that earlier and extended treatment provides the best outcomes.”
Clark Tedford, PhD, President and CEO of LumiThera, summarized the broader implication: “The extended trial results demonstrate that Valeda is potentially modifying the trajectory of vision loss in dry AMD patients and can offer sustainable benefits over several years with continued treatment.”
Take the Next Step
The LIGHTSITE clinical program represents some of the most meaningful progress in dry AMD research in recent history. For patients who have been told that dry AMD is simply something to monitor and manage, these results offer a different message: early intervention with Valeda can improve your vision and may change the course of your disease.
If you have been diagnosed with dry AMD, this is the time to explore your options. Our team is here to answer your questions and help you decide whether Valeda is right for you.
To get started, email us at [email protected], call us at 858-451-1911, or fill out our Valeda inquiry form here: https://www.rcsd.com/valeda.
We will reach out within 24 hours to guide you through next steps and answer any questions you may have.
